A 7 page booklet that students can take with them and monitor their own competencies throughout the course.
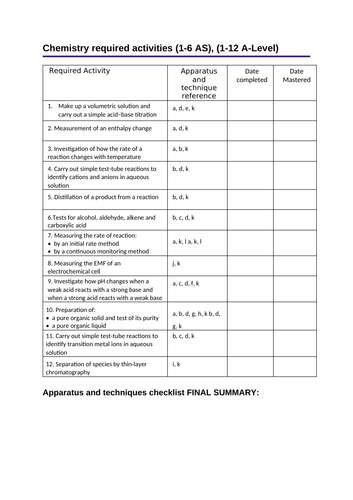
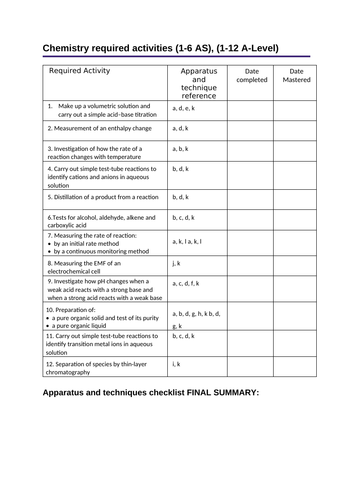
Page 1: Apparatus and techniques checklist summary (to be filled in last)- students can mark off date completed and date mastered
Required Activity
Apparatus and technique reference
Date completed
Date Mastered
Page 2: Apparatus and techniques checklist - explaining each technique
Page 3: Apparatus and techniques in detail monitoring log -
Apparatus and techniques
Phase 1: Demonstrate
Phase 2: Practise with support
Phase 3: Practise without support
Phase 4: Fluent
Page 4: Common Practical Assessment Criteria in A-level Chemistry: explaining each CPAC
Page 5-6: Monitoring practical competencies:
Date
Practical
Checkbox for each competency
Page 7: AS Assessed Practical’s: Preparation work required and when it will be taught in the course
Enjoy
Something went wrong, please try again later.
This resource hasn't been reviewed yet
To ensure quality for our reviews, only customers who have purchased this resource can review it
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.