

In this lesson we go over the subatomic particles in the atom and the concept of the nucleus. This is lesson four in our physical chemistry series for Unit 2: Atomic Structure (from the Cambridge International AS Chemistry Curriculum (9701) 2019-2021 curriculum).
LESSON OBJECTIVE: Understand the properties of subatomic particles of an element including naming, mass and charge conventions for elements and isotopes.
LEARNING OUTCOMES (from the Cambridge AS Chemistry Curriculum 2019-2021):
2.1 Particles in the atom
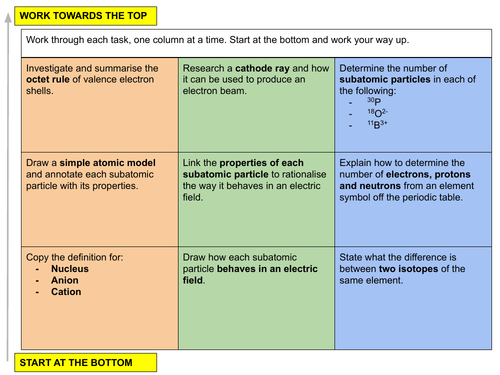
a) identify and describe protons, neutrons and electrons in terms of their relative charges and relative masses
b) deduce the behaviour of beams of protons, neutrons and electrons in electric fields
c) describe the distribution of mass and charge within an atom
d) deduce the numbers of protons, neutrons and electrons present in both atoms and ions given proton and nucleon numbers (atomic and mass numbers) and charge
2.2 The nucleus of the atom
a) describe the contribution of protons and neutrons to atomic nuclei in terms of proton (atomic) number and nucleon (mass) number
b) distinguish between isotopes on the basis of different numbers of neutrons present
c) recognise and use the symbolism xyA for isotopes, where x is the nucleon (mass) number and y is the proton (atomic) number
Something went wrong, please try again later.
This resource hasn't been reviewed yet
To ensure quality for our reviews, only customers who have downloaded this resource can review it
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.