

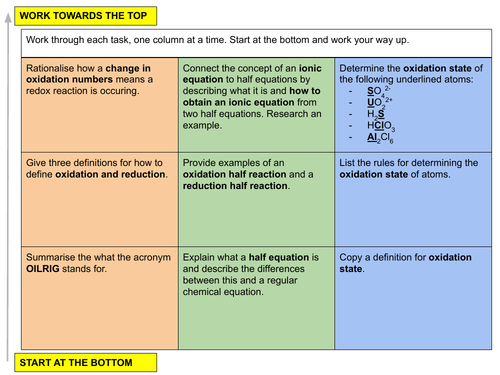
In this lesson we discuss the concept of redox processes from reduction and oxidation reactions, half equations, ionic equations and how to determine oxidation states (oxidation numbers). This is lesson eighteen in our physical chemistry series for Unit 6: Electrochemistry (from the Cambridge International AS Chemistry Curriculum (9701) 2019-2021 curriculum).
LESSON OBJECTIVE: Understand and explain redox reactions in terms of electron transfer and oxidation numbers.
LEARNING OUTCOMES (taken from the Cambridge International AS and A Level Chemistry (9701) 2019-2021 curriculum):
6.1 Redox processes: electron transfer and changes in oxidation number (oxidation state)
a) calculate oxidation numbers of elements in compounds and ions
b) describe and explain redox processes in terms of electron transfer and changes in oxidation number
Something went wrong, please try again later.
This resource hasn't been reviewed yet
To ensure quality for our reviews, only customers who have downloaded this resource can review it
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.