












AQA GCSE 9-1 CHEMISTRY UNIT 3.2 Amount of substance HIGHER moles, limiting reagent
4.3.2 Use of amount of substance in relation to masses of pure substances
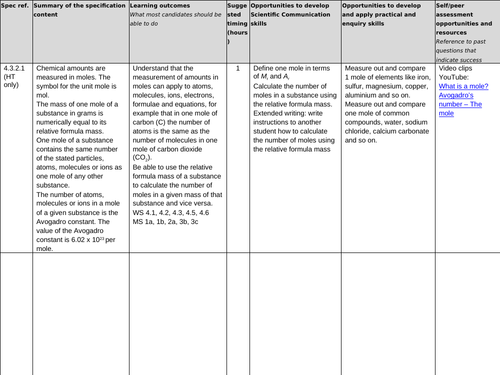
4.3.2.1 Moles (HT only)
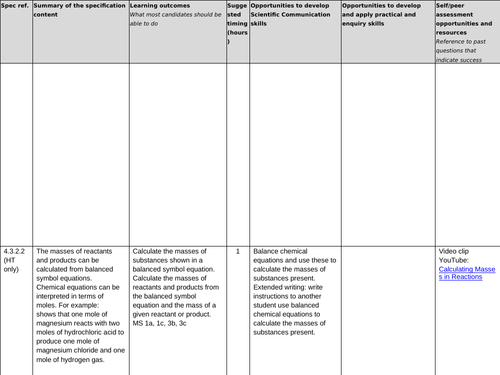
4.3.2.2 Amounts of substances in equations (HT only)
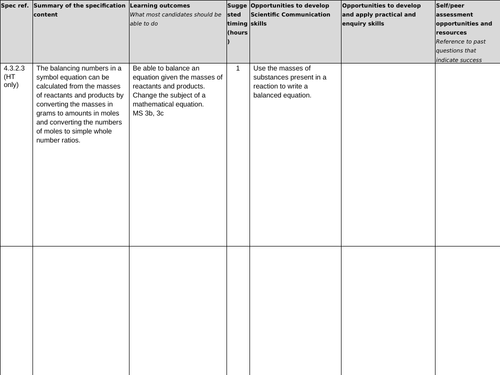
4.3.2.3 Using moles to balance equations (HT only)
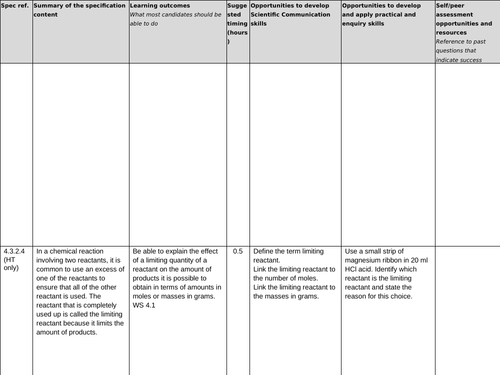
4.3.2.4 Limiting reactants (HT only)
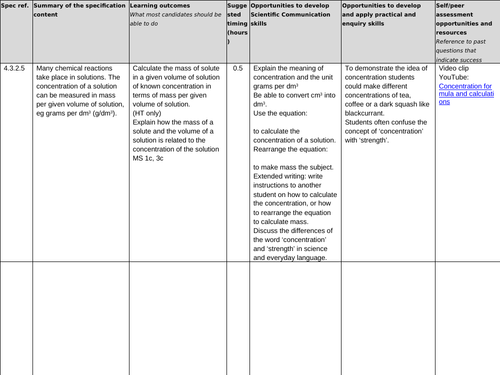
4.3.2.5 Concentration of solutions
Content split over 6 lessons (lessons in our school are 40 minutes so can condense material for longer lessons if required)
All exam questions have been removed for copyright purposes
All extension questions available on each slide
Answers all underneath each slide
Support also available where necessary
AfL sections and mini quizzes
Reducing the need for photocopying
Homework
Homework can also be used as extension sheets in lessons - or for higher ability students
Lesson 1 & 2: Moles basics
To understand the terms molar mass and mole
To know that moles are the unit for amount of substance and to be able to use the molar equation
To be able to calculate the number of atoms in elements and molecules
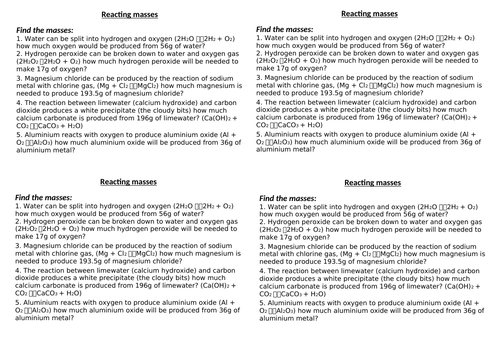
Lesson 3: Reacting masses with moles
To explain balanced symbol equations in terms of moles
To be able to calculate the amount of product from a given reactant
To consolidate learning with exam questions
Lesson 4: Balancing equations with moles
To complete the moles recap starter
To understand how to deduce the stoichiometry of a reaction from the masses of the reactants and products to enable us to write the balanced equation
To consolidate learning with further questioning
Lesson 5 & 6: Limiting reagents
To understand the term ‘limiting reagent’
To be able to identify the limiting reactant in a reaction
To be able to calculate the mass of the reactant from the limiting reactant using moles
Lesson 7: Concentration of solutions g/dm-3 (no moles)
To be able to define the term ‘concentration’ (H)
To be able to convert between cm3 and dm3
To understand how to calculate the concentration of solutions in g dm-3 or g/dm3
Get this resource as part of a bundle and save up to 26%
A bundle is a package of resources grouped together to teach a particular topic, or a series of lessons, in one place.
Something went wrong, please try again later.
This resource hasn't been reviewed yet
To ensure quality for our reviews, only customers who have purchased this resource can review it
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.
