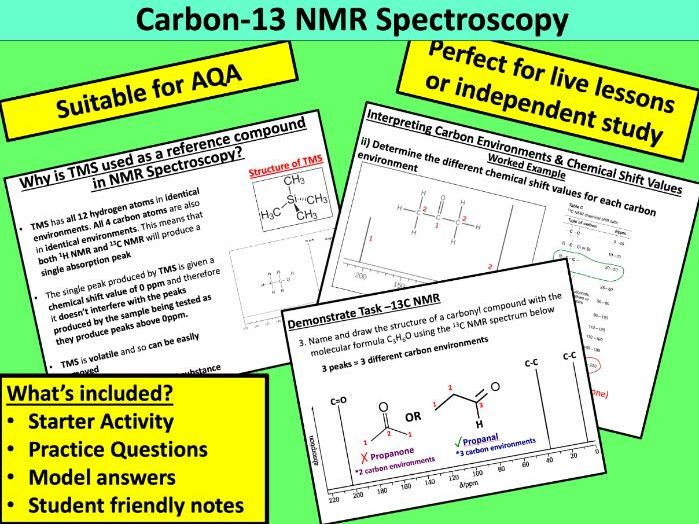
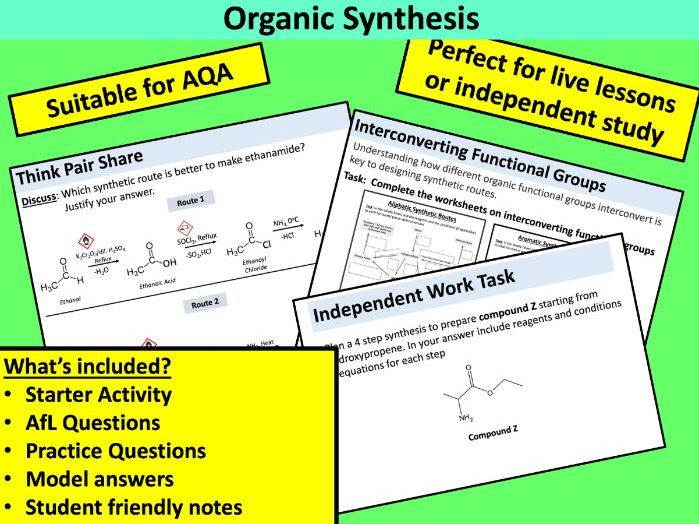
Teach Science & Beyond
Over 300 resources available for KS3-KS4 Science, KS5 Chemistry and Whole School! Lesson resources are suitable for live lessons in school, remote teaching at home or independent student study. It’s your choice how you use them 😊 Don’t forgot to explore my free resources too!