124Uploads
32k+Views
8k+Downloads
Chemistry

C1. 8 electronic structures
Aimed at a mixed ability year 9 class
Starter Activity
Exam question starter – go through and green pen
Main –
Recap where to find the number of electrons an atom has, go through electronic structure rules. Do a few examples on the board
Two worksheets – first worksheet students need to find out the number of protons, neutrons and electrons each element has, the second they need to draw the electronic structures
Extension: explain why sodium and lithium might react in a similar way
Write a step by step guide for somebody to explain how you draw electronic structures
Plenary
Exam question plenary – spot mistakes in the electronic structures

C3.3 Ionic bonding - foundation
Aimed at a lower ability class
Starter Activity
Ions recap, what charge do elements have in different groups
Stretch question to think about how group 1 and 2 will interact with group 6 and 7
Main –
Go through the video and answer the questions, go through answers
Practice drawing ionic compounds Extension: explain the difference between KBr and K2O
Go through the answers which are on the slides
Exam question practice to move onto if have a chance
Plenary
4 mark exam question – peer assess
Good to show the detail they need in their answers

C3.2 atoms into ions - Foundation
aimed at a lower ability class
Starter Activity
Identify the atom on the board
Stretch – how do you know which atom it is
Main –
Recap how to find out electron/proton/neutron numbers and how to draw electron configuration. Think pair share, what can the two atoms do to have full outer shells
Students explain in their books what a positive and negative ion is
3 quick questions – green pen mark
Go through rules so we always know the charges of common ions
Worksheet to complete – label periodic table with rules and then practice drawing ions
Extension: describe and explain how ions are formed
Plenary
Exam question plenary – peer assess

C3.5 covalent bonding - foundation
aimed at a lower ability class
Starter Activity
Mark homework
Main –
Think pair share – spot the difference between ionic and covalent bonding. Then write down definition of covalent bonding
Watch video, only watch until 2 minutes. Answer questions whilst video is going. Goo through and green pen answers after
Go through the two examples on the board and show what happens
Students then have a go at completing their worksheet to show covalent bonds
Extension: decide if the compounds are ionic or covalent and explain how they know
Plenary
Plenary, fill in the gaps and go over answers

C3.4 Giant ionic structures - foundation
aimed at a lower ability class
Starter Activity
Using previous knowledge make predictions about ionic compounds
Main –
Go through and discuss giant ionic lattices
Students move around the room to find the information sheet to find out if there prediction was correct or not and why
Practical with a dissolved ionic compound to show it can conduct electricity. Make sure say that molten ionic compounds too and what a molten ionic compound is
Plenary
Exam question plenary

C12. treating waste water
For low ability students. targets level 4
Starter: differentiated stater based on prior learning of the topic
answers on following slide
Main: think pair share: where do we get waste water from
video: watch a video, try and pick out what the stages of treatment for waste water are
differentiated worksheets, to explain the process of waste water treatment
Extension: differentiated extension questions
Plenary: copy and complete two sentences
Bundle

C11 - The earths atmosphere
All resources aimed at foundation students
C11.1 + C11.2 combined into one lesson
C11.3 greenhouse gases
C11.4 global climate change
C11.5 atmospheric pollutants

C3 Quantitative chemistry tutoring
A tutoring powerpoint I used does not include percentage yield.
Comes with a key question document and exam questions.
All answers to powerpoint included as animations or in the notes below
Bundle

Separation techniques KS3
Bundle of lessons for year 8
L1 - elements compounds mixtures
L2 - Dissolving
L3 - Solubility
L4 & 5 - filtering and evaporating
L6 - Melting points
L7 - Simple distillation
L8 - Fractional distillation
L9 - Chromatography

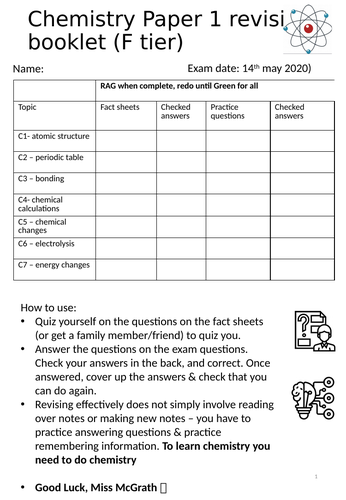
Paper one chemistry revision
Complete paper one chemistry revision
Inspired by Dr Edmunds
Each section has a fact sheet to fill in, page numbers are highlighted from the foundation revision CGP booklet. Facts are based on the level 4 requirements from the kerboodle scheme of work.
The booklet contains the answers to the fact sheets
there are then practice questions with answers for each section