497Uploads
167k+Views
71k+Downloads
All resources

OCR AS Chemistry: Representing the formulae of Organic Compounds
OCR AS Chemistry: Formulae for Organic Compounds
This PowerPoint is a whole lessons included with student activities, animated answers, homework questions with answers provided.
This lesson covers:
molecular formula
empirical formula
general formula
displayed formula
structural formula
skeletal formula

GCSE Physics: Electrical Power and Work Done
This presentation covers OCR Gateway Physics 9-1 P7.2.1 Electrical Power and Work Done. All presentations come with student activities and worked solutions.
Definition of power
Converting between W and kW
Converting between seconds, minutes, and hours
Calculating work done in kWh and J
Converting between kWh and J

OCR AS Chemistry: The Chemistry of Haloalkanes
OCR AS Chemistry: The Chemistry of Haloalkanes
This PowerPoint is a whole lessons included with student activities, animated answers, homework questions with answers provided.
This lesson covers:
Naming Haloalkanes
Classifying Haloalkanes (primary, secondary, tertiary)
Electronegativity
Reaction mechanism for hydrolysis
Rates of reactions for hydrolysis
Reaction conditions for hydrolysis

GCSE Physics: Electricity and Subatomic Particles
This PowerPoint presentation with worked examples and student questions covers:
• Atoms and the particle model for solids, liquids and gases
• Subatomic particles and the property of charge
• Structure of a metal
• Explanations of the Van de Graff and Lightning.

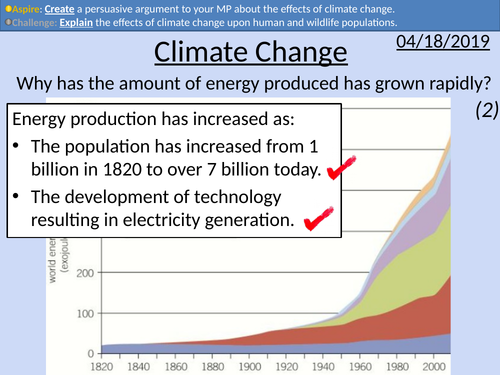
GCSE Physics: Climate Change
This PowerPoint presentation with worked examples and student questions covers:
• Types of greenhouse gases – carbon dioxide, methane, nitrous oxide, and water vapour.
• Greenhouse effect with activity
• Class discussion on news report of effects of climate change
• Extended writing task with student friendly mark scheme and scaffolding
• Data analysis task
• Explanation of data collection of CO2 levels

GCSE Physics: Constructing Circuits
This PowerPoint presentation with worked examples and student questions covers:
• Constructing Series Circuit
• Constructing Parallel Circuits
• Analysing results for Potential Difference
• Analysing results for Current
• Clear resource list, diagrams and photos to aid teaching.

GCSE Chemistry: Producing Electricity Using Chemistry
This PowerPoint presentation with worked examples and student questions covers:
Chemical cells uses
Fuel cell uses
Comparing fuel cells and chemical cells
Environmental impact of fuel cells and chemical cells
The structure of fuel cells
The operation of fuel cells
Half-equations for fuel cells

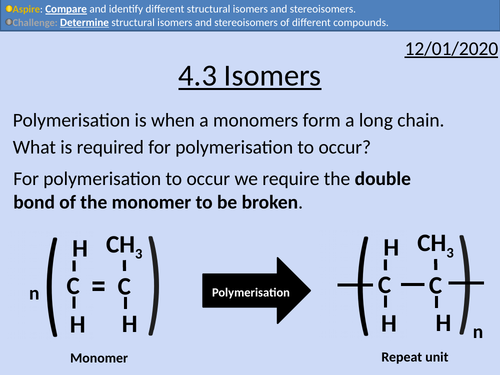
OCR Applied Science: 4.3 Isomers
This PowerPoint presentation with worked examples and student activities covers: Topic 4.3 of Module 1: Science Fundamentals of the OCR Applied Science Spec.
• Stating definitions and comparing structural isomers and stereoisomers.
• Condensed structural formula
• Lines of symmetry for structural isomers
• Cis- and Trans isomers
• Optical isomers as non-superimposable mirror images.
• Wedge and Dash Notation
• Identifying chiral centres (asymmetric carbons)
• Le Bel-van’t Hoff rule
• Determining the maximum number of isomers.

OCR Applied Science: 21.2.1 Types of Testing
OCR Applied Science Level 3 - Module 21: Product Testing Techniques.
This PowerPoint presentation with worked examples and student activities covers: Topic 2.1 of Module 21: Product Testing Techniques.
2.1 Types of testing i.e.:
• in-vitro
• in-vivo
• titration
• extraction and separation

OCR Applied Science: 21.2.2 Testing During Development
OCR Applied Science Level 3 - Module 21: Product Testing Techniques.
This PowerPoint presentation with worked examples and student activities covers: Topic 2.2 of Module 21: Product Testing Techniques.
2.2 Laboratory testing during development i.e.:
• formulation
• production
• quality control and assurance
• after sale monitoring.

OCR Applied Science: 4.2 Polymers and Carbon Compounds
This PowerPoint presentation with worked examples and student activities covers:
Topic 4.2 of Module 1: Science Fundamentals of the OCR Applied Science Spec.
Determining the empirical formula for compounds
Draw monomers and repeat units using structural and skeletal formula of the following polymers:
Polyethene
Polypropene
Polylactate
Polystyrene
Polyvinyl chloride (PVC)

OCR Applied Science: 21.2.3 Effectiveness of Tests
OCR Applied Science Level 3 - Module 21: Product Testing Techniques.
This PowerPoint presentation with worked examples and student activities covers: Topic 2.3 of Module 21: Product Testing Techniques.
2.3 Effectiveness of test
• Appropriate test method
• Data collection validity and reliability
• Consistent chemical composition
• Hazards and risks of use

OCR Applied Science: 21.1 Regulatory Bodies
This PowerPoint presentation with worked examples and student activities covers: Topic 1.1 and 1.2 of Module 21: Product Testing Techniques.
Understand the influence of regulatory bodies on development of consumer products.
1.1 The relevant governing bodies that oversee product safety for
manufacturers and consumers of products.
1.2 How governing bodies influence how quality control is applied.
Bundle

OCR AS level Chemistry: Organic Synthesis
OCR AS level Chemistry: Organic Synthesis is apart of the Module 4: Core Organic Chemistry and Analysis
All presentations come with worked examples, solutions and homeworks
Heating under reflux
Distillation
Re-distillation
Purifying Organic Products
Removing impure acids from organic compounds
Drying agents
Functional Groups - Alkane, Alkene, Haloalkane, Alcohols, Carboxylic Acid, Ketone, Aldehyde, Ester, Amine, Nitrile.
One-step synthetic routes with reagents and conditions
Two-step synthetic routes with reagents and conditions
Bundle

OCR AS level Chemistry: Alkanes
OCR AS level Chemistry: Alkanes is apart of the Module 4: Core Organic Chemistry and Analysis
All presentations come with worked examples, solutions and homeworks
Bundle

OCR AS level Chemistry: Haloalkanes
OCR AS level Chemistry: Haloalkanes is apart of the Module 4: Core Organic Chemistry and Analysis
All presentations come with worked examples, solutions and homeworks
Naming Haloalkanes
Classifying Haloalkanes (primary, secondary, tertiary)
Electronegativity
Reaction mechanism for hydrolysis
Rates of reactions for hydrolysis
Reaction conditions for hydrolysis
Definitions for CFC (Chlorofluorocarbons) and HCFC (Hydachlorofluorocarbons)
Creation of ozone
Depletion of ozone with CFCs
Reaction steps including initiations and propagation
Bundle

OCR AS level Chemistry: Alcohols
OCR AS level Chemistry: Alcohols is apart of the Module 4: Core Organic Chemistry and Analysis
All presentations come with worked examples, solutions and homeworks
Naming alcohols
Classifying alcohols (primary, secondary, tertiary)
Electronegativity
Polar and non-polar molecules
Explaining physical properties of alcohols compared to alkanes
Volatility
Solubility
Melting points
Chain length and London forces
Combustion of alcohols
Reflux condition for reactions
Primary alcohol to aldehydes
Primary alcohols to carboxylic acids
Secondary alcohols to ketones
Dehydration of alcohols
Substitution reactions for alcohols

GCSE Biology: Photosynthesis Experiments
This two lesson presentation covers OCR Gateway Biology 9-1 B1.4.2 Photosynthesis Experiments
This PowerPoint is a whole lessons included with student activities and animated answers.
The positive test for starch
Experimental procedure for testing starch in leaves.
Testing for the need of chlorophyll for photosynthesis.
Testing for the need of light for photosynthesis.
Testing for the need of carbon dioxide for photosynthesis.
Testing oxygen produced via photosynthesis.
Testing light intensity affects photosynthesis.

OCR Applied Science: 6.3 Electrical Properties
This PowerPoint presentation with worked examples and student activities covers:
Topic 6.3 of Module 1: Science Fundamentals of the OCR Applied Science Spec.
Current as flow of charge in a conductor.
Use the equation: I = ΔQ ÷ Δt
Ohm’s law illustrates the relationship of V ∝ I
Use the equation: potential difference (V) = current (A) × resistance
Use the equations for adding resistors in series and parallel
Compare electromotive force and potential difference
Use the equation: charge © = current (A) × time (s)
Use and recognise the equation for mean drift velocity
Use the equation: energy transferred (work done) (J) = charge © × potential difference (V)
Use the equation: energy transferred (J, kWh) = power (W, kW) × time (s, h)
Use the equation: power (W) = energy (J) ÷ time (s)

OCR Applied Science: 2.2 Reactions
This PowerPoint presentation with worked examples and student activities covers:
Topic 2.2 of Module 1: Science Fundementals of the OCR Applied Science Spec.
Oxidation and reduction (redox) reactions
Addition reactions of alkenes to include full balanced symbol equations
Substitution reactions of alkanes and haloalkanes to include full balanced
equations
Addition polymerisation to include identification of monomers and repeating units
Condensation polymerisation to include identification of monomers and repeating units
Definition of a radical
The role played by UV light in producing chlorine radicals from CFCs in the
depletion of the ozone layer
Equations to show how chlorine radicals can destroy many ozone molecules
Displacement reactions to include full balanced equations for metals and halogens.